|
However, antigen display on OMVs can be difficult to control and highly variable due to bottlenecks in protein expression and localization to the outer membrane of the host cell, especially for bulky and/or complex antigens. The data provided information on the complexity of heterologous expression of AT antigens at the OMV surface and suggested that optimal expression strategies should be developed on an antigen-to-antigen basis.Įngineered outer membrane vesicles (OMVs) derived from Gram-negative bacteria are a promising technology for the creation of non-infectious, nanoparticle vaccines against diverse pathogens. Next, we investigated whether a similar chimeric surface display strategy could be applied to other AT antigens, i.e., secreted fragments of Prn (aa35-350) of Bordetella pertussis and VacA (aa65-377) of Helicobacter pylori. coli AT HbpD-fusions of chimeric constructs we improved surface display and successfully generated Salmonella OMVs decorated with a secreted and immunogenic PmpD passenger fragment (aa68-629) to 13% of the total protein content. OMVs are considered safe vaccine vectors well-suited for mucosal delivery. trachomatis full-length (FL) PmpD, as well as truncated PmpD passenger constructs fused to a “display” autotransporter (AT) hemoglobin protease (HbpD) and studied their inclusion into outer membrane vesicles (OMVs) of Escherichia coli and Salmonella Typhimurium. 
In this study, we explored the expression of C. To combat the global spread of asymptomatic infection, development of effective (mucosal) vaccines that offer both systemic and local immune responses is considered a high priority. The percentage of HbpD(Δd1)-SpT ligated with Sp圜atcher-SnoopCatcher is plotted as a function of time.Ĭhlamydia trachomatis is the bacterial pathogen that causes most cases of sexually transmitted diseases annually. 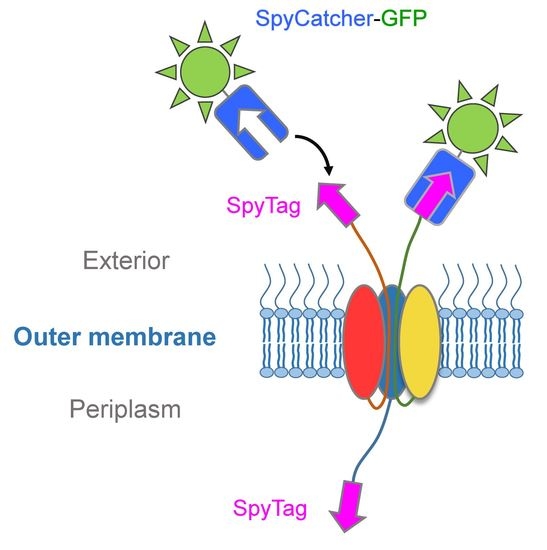
(D) Quantification of ligation efficiency to HbpD(Δd1)-SpT as in panel B. The sizes of the molecular mass markers are indicated on the left side of the panel. A proteolytic product of the adduct is indicated with an asterisk. HbpD(Δd1)-SpT and the adduct are indicated on the right-hand side of the panel. (C) SDS-PAGE and Coomassie staining of reaction mixtures containing a 4:1 molar ratio of Sp圜atcher-SnoopCatcher to HbpD(Δd1)-SpT incubated for 1, 2, 4, 8, 16, or 24 h at 4☌. The percentage of HbpD(Δd1)-SpT ligated with Sp圜atcher-SnoopCatcher is plotted as a function of the molar ratio Sp圜atcher-SnoopCatcher:HbpD(Δd1)-SpT. The intensities of the adduct and HbpD(Δd1)-SpT in the gel shown in panel A were determined by densitometry. (B) Quantification of ligation efficiency to HbpD(Δd1)-SpT. 
Reactant proteins, the adduct, and the major outer membrane protein OmpA are indicated on the right-hand side of the panel. (A) SDS-PAGE and Coomassie staining analysis of reaction mixtures of OMVs harboring HbpD(Δd1)-SpT and SpC-SnC incubated for 21 h at 4☌. 
Ligation of OMVs displaying SpyTag using increasing amounts of Sp圜atcher-SnoopCatcher (SpC-SnC).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
